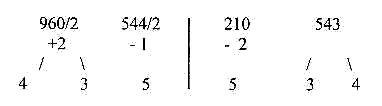|
9. Chemical aspects on the distribution of
codons
|
1. Glycolysis and Citrate cycle: derivation of
the amino acids:
References:
P.O.Karlson: "Biokemi", 1976.
D.E.Nicholson:
"Metabolic Pathways", 1976.
Amino acids from the different
stations:
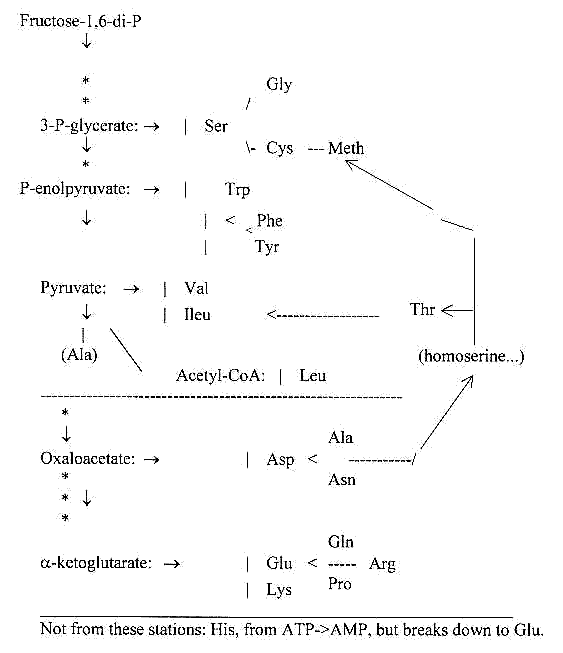
There are obviously two simple
ways to Ala,
from Pyruvate and from Oxaloacetate
There is a bit unsure too, from which stations
one ought to refer Thr and Meth, along the "side way"
via homoserine from Oxaloacetate?
|
2. Mass number (A) distribution for R-chains of the
24 amino acids, 6 stations:
Triplet numbers of a dimension
chain: : 543 + 210 = 753
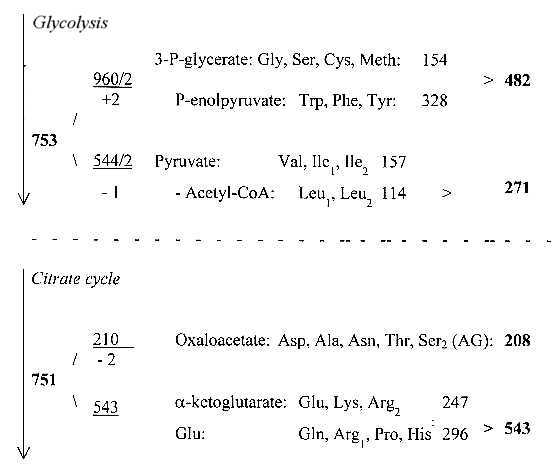
From the loop back from Oxaloacetate via Homoserine Thr is derived,
which can transform into Ileu.
This same loop connects Meth: Meth <==>
Cys --> Ser --> Gly. That is why Ser2 has been seen as derived
from Oxaloacetate here.
With these options we have a halving (+/-1) of the total mass
of ams, one half from glycolysis, one half from the citrate cycle
inside the mitochondrias.
Number of amino acids and A-sums in the groups above:
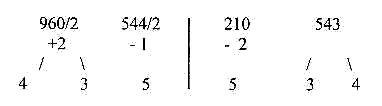
According to the numbers we could probably also see the derivation
way of ams in the opposite direction: from the key substance a-ketoglutarate,
the C5-substance responsible for aminating the amino acids, towards
oxaloacetate and outwards, to C4 and C3 substances.
Another key substance in the citrate cycle is Succinyl-Coa, which
follows directly after a-ketoglutarate
and leads to porphyrin rings: it could be said to mark the crossing
point between the vegetable kingdom and the animal kingdom, between
the Photosynthesis and the Glycolysis. We could presume that it
marks a central point in a bigger loop or dimension chain.
We can also see the way from Pyruvate to a-ketoglutarate
as a way of synthesis, that is directed inwards (as into the mitochondrias
too), also meaning inwards higher dimension degrees in a chain.
|
3. Codons with U-base in 1st and/or 2nd position:
We should notice that all ams but Gly derived from stations in
the glycolysis have the U-base in 1st or 2nd position of their
codons, if we disregard the possibility of Ala deriving from Pyruvate;
no ams from stations in the citrate cycle.
This circumstance could possibly have connection
with the fact that U (and T) as coenzymes (-TP, -DP-, MP-forms)
are the active ones in syntheses or break down of carbohydrates.
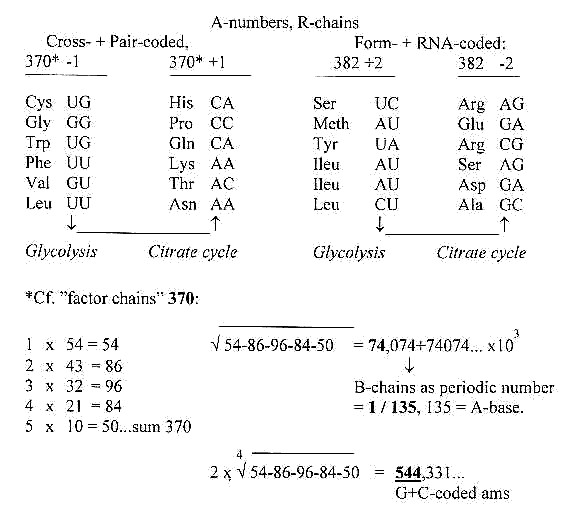
Cf. numbers 27 and 8: √27/8 ---> inverted =
544,33105.. x 10^-1 in list-file 13.
|
4. Functional groups / "endings" in R-chains of
the the amino acids:
(Or typical atom groups.)
Besides the list above and the comment on the U-base codons,
there hasn't been possible to find anything indicating a connection
between chemical aspects of the individual amino acids and the
distribution of codons. (This according to the scientists.) Collective
mass numbers seem more governing, according to the papers here.
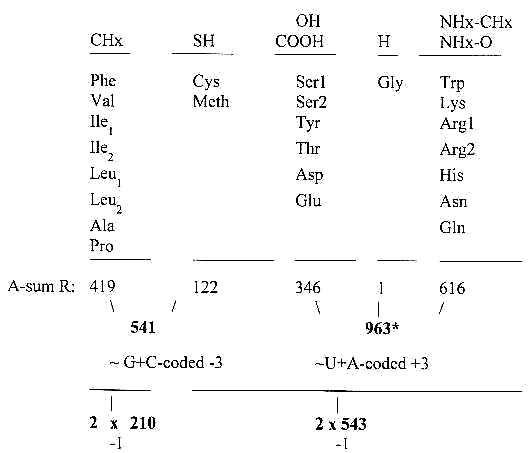
This grouping, differentiating only the nonpolar group CHx from
the other ones, connects to the triplet series 2 x (543 + 210)
It could eventually reveal one figuration in the fairy dance of
the amino acids ? (Cf. the position of C in relation to N and
O on the page about "A-Z-"numbers.)
*543 — 432 + 321 + 210
963
(If Gly is put together with the nonpolar group we get 2 x 210,
2 x 543, -2.)
*
|
© Åsa Wohlin:
Free to distribute if the source is mentioned.
Texts are mostly extractions from a booklet series, made publicly available
in year 2000.
|
Links and Notes
To the background
model
Table
24 ams A, Z, N
Abbreviations
- ways of writing -
List
on 17 short files
The
17 files
as one document, pdf
Contact:
u5d
Latest updated
2022-09-29
|